 If ∆S is negative, then the negative signs (from the subtraction and the sign of ∆S) will cancel out, and so as T∆S gets bigger, ∆G will get more positive. T is always positive, so if ∆S is positive then a bigger T∆S will make ∆G more negative (since we subtract T∆S). As T increases, the T∆S component gets bigger. ∆H is still positive and ∆S is still whatever sign you figured out above. Since ∆H and ∆S don't change significantly with temperature (given in the question), we can assume that they keep the same signs and values: i.e. The entropy increases as the freedom of movement of molecules increases with phase change. If ∆G is negative (from the question), is the reaction spontaneous or non-spontaneous?Ģ) Let's use ∆G = ∆H - T∆S again. It is the increase in entropy when a solid melt into liquid. 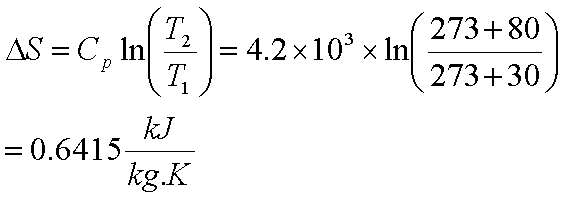
From these values, we can know for certain whether ∆S is positive or negative (hint: remember that we are subtracting ∆G!).ġ) Knowing the sign of ∆G is enough to say whether the reaction is spontaneous or not under these conditions. Temperature is always positive (in Kelvin). Thermodynamics Entropy Change Entropy Change What is Entropy The decrease in enthalpy may contribute to the spontaneity of a process but it cannot explain spontaneity in all the cases. 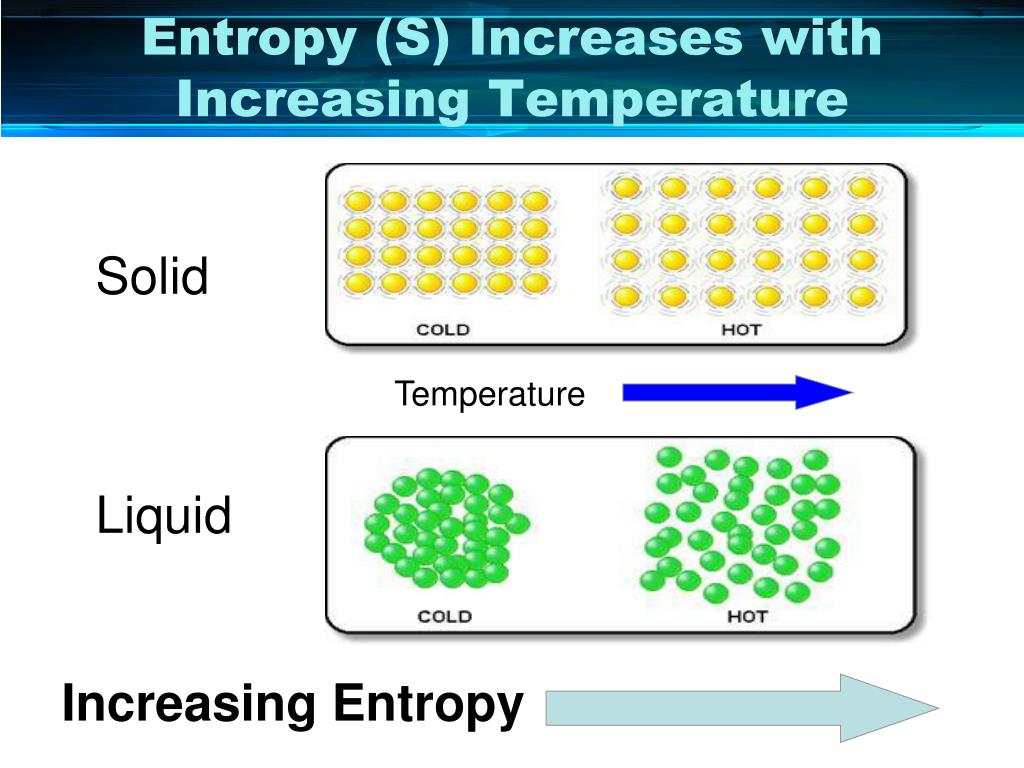 
We know (from the question) that ∆G is negative and that ∆H is positive. This looks like a homework question, so I'll give you some hints to get you on the riht path rather than answering directly.ģ) We know that ∆G = ∆H - T∆S. This equation is exciting because it allows us to determine the change in Gibbs free energy using the enthalpy change, H Delta text H H delta, start text, H, end text, and the entropy change, S Delta text S S delta, start text, S, end text, of the system. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
